In April, ZoomRx collaborated with Dr. Farrah Mateen and her research team at the Massachusetts General Hospital Department of Neurology to understand how neurology departments across the United States and Canada were rapidly adapting their clinical care delivery systems to minimize risk of SARS-CoV-2 transmission.
The study surveyed 250 multiple sclerosis (MS) treating neurologists and 192 neuromyelitis optica spectrum disorder (NMOSD) treating neurologists in the U.S. and Canada from April 14 to May 3, 2020. The results of this study were recently published in the Journal of Neurology and the Journal of Neuroimmunology, respectively.
At the time of publication, uncertainty around the potential neurological manifestations of SARS-CoV-2 and the increased risk for patients on immunosuppressive therapies (ISTs) abounded. The research highlighted the urgent need for streamlined and comprehensive treatment guidelines.
“Clarity on their [patients] risk, which is probably elevated compared to the general population, but unclear to what extent and if, in particular, associated with certain immunosuppressant drugs.” - Physician discussing unmet needs
Neuroimmunology patients receiving high-efficacy ISTs are believed to have a higher risk of becoming infected with SARS-CoV-2 or developing a severe case of COVID-19. Conversely, some physicians have hypothesized that ISTs may be therapeutically relevant due to the systemic hyper-inflammation seen in severe COVID-19 cases (Pharmacological Research).
The MS publication hypothesized that with increased prescribing complexity, the COVID-19 pandemic could dampen enthusiasm for induction therapy (early use of higher-efficacy therapies). Physicians noted that the most commonly avoided agents for initiation were high-efficacy ISTs: alemtuzumab, cladribine, ocrelizumab, rituximab and natalizumab. Risk-averse physicians and patients may instead favor a traditional escalation approach (early use of lower efficacy therapies), thus inciting a potential renaissance for lower-efficacy injectables.
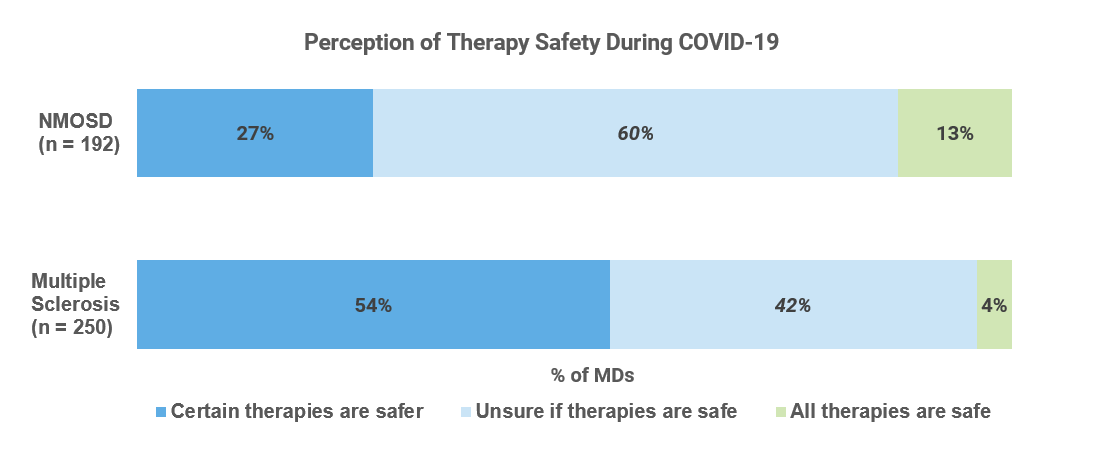
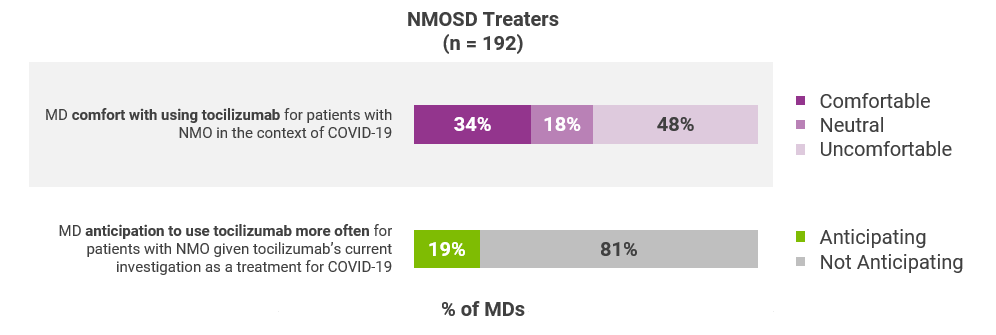
In the NMOSD publication, the need for comprehensive guidelines was particularly evident. Neurologists noted different and sometimes diverging stances on the treatment of NMOSD patients with tocilizumab, a IL-6 inhibitor that was evaluated as a treatment for COVID-19.
In both the MS and NMOSD research, responses underscored similar barriers to patient care: technological and reimbursement challenges with telemedicine, inadequate home infusion capabilities, and the economic impact on an already financially-burdensome disease. Studies have since emerged that detail how neurology departments moved swiftly and vigilantly in March and April to create virtual care continuums and highlighted challenges so that others could adapt their practices [Neurology].
“The physician reimbursement for phone/virtual visits are less than face-to-face visits although we are still doing mostly the same work including prescriptions, requisitions, forms etc.”
“Due to the need for distancing in the infusion room and the pre-COVID volume of infusions that must be maintained, we are having challenges to keep patients on their infusion schedule. Home care services are also limited. The discussion about DMT selection is difficult to do by phone but that is the only option for some patients."
"We need guidance on safety of immune suppression. We also need better financial and administrative support to switch patients to home infusions, where appropriate.”
Since this research was conducted...
Since research was completed in early May, SARS-CoV-2 neurological manifestations have been well-characterized with symptoms ranging from dizziness and headaches to myositis and encephalopathy. However, whether there is a direct viral infection of brain tissue by SARS-Cov-2 versus a generalized systemic response remains uncertain (Therapeutic Advances in Neurological Disorders). Due to the increased instances of cerebrovascular and cognitive manifestations, neurological consultations are increasingly recommended in the care of COVID-19 patients. Consequently, increased strain is placed on an already time-constrained physician population.
National and international guidelines have been published and generally recommend continuation of MS and NMOSD therapies; however, these guidelines do not reach a consensus on patient management, as noted recently in a Nature publication.
Final Thoughts
As the pandemic continues to persist in the United States, there is likely an increased risk of future MS relapses or NMOSD attacks due to alterations in disease surveillance and treatment approaches. Additionally, it’s worth noting that studies have historically suggested that viruses (VZV, EBV, HHV-6, etc.) may serve as environmental triggers for MS and NMOSD pathogenesis (CNS and Neurological Disorders Drug Targets). While there is not strong evidence to support milder coronaviruses as potential environmental triggers, given that SARS-CoV-2 is a novel and more severe coronavirus, it will be important to monitor the incidence rates of neuroimmunological diseases following the pandemic.
Have questions about this research? Please reach out below and a member of our team will follow-up.
Learn more.



